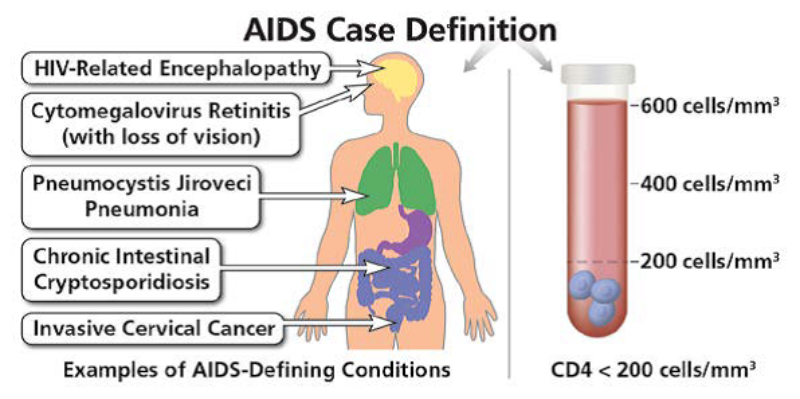
The prospect of having HIV may provoke fear and dread in the sexually active. This anxiety is reasonable given the history surrounding HIV and AIDS in the United States and globally. HIV infection is a profoundly serious medical condition, though not the sentence of an agonizing wasting death that it was during the 1980s and 1990s. There are now medications that can slow the progression of the disease by decades, in many cases, giving an extension of life to those diagnosed with this progressive lethal virus rather than the near specter of a wasting, suffering death.
The question remains as to why so many in the United States avoid HIV testing and prevention when it is so important to avoid or at least become aware of an infection early. It is allowing early treatment with the potential to slow or avoid the fate they see that is being portrayed by the news media visiting impoverished AIDS-stricken countries in Africa's west, central, and southern regions where a diagnosis of HIV has been the equivalent of a death sentence (Senate Foreign Relations Committee [SFRC], 2023). Perhaps it is the fear of receiving a diagnosis with a fatal infectious disease, or the fear of living with the responsibility of keeping a demanding disease such as HIV/AIDS under control. Keeping HIV under control is a time and resource-consuming task. However, the option of letting the disease run its course unchecked would be an extremely poor choice, as well.
Whatever factors bring hesitation to those needing HIV prevention counseling, screening, or treatment, we know that one commonly held fear is the stigma attached to having HIV or AIDS. Having HIV brings a "scarlet letter" fear of repercussions due to possessing an unwanted health status. When questioned, many of those newly diagnosed with HIV seem unaware that federal laws exist to protect them from discrimination and ensure that benefits such as any social or medical services they would otherwise qualify for will not be withheld. As healthcare professionals, it is important to share with those in high-risk groups that key legislative pieces exist that offer protection from discrimination. Interestingly, some of the mandates and pieces of legislation protecting those with current needs predate the HIV/AIDS epidemic, e.g., Section 504 of the Rehabilitation Act of 1973 and pieces of the 1990 Americans with Disabilities Act (ADA) (HIV.gov, 2025c). These well-established laws mean services that apply to an otherwise qualified individual will also be available to HIV-infected individuals.
Title II of the ADA prohibits discrimination by state and local government organizations, even those not dependent on federal funding. For those nervous or fearful concerning how those close to them might be treated should they acquire HIV, Title II of the ADA also protects an HIV positive individual's friends and family against discrimination or denial of services that might come from being related to someone with HIV (HIV.gov, 2025c).
Information privacy regarding an individual's HIV status, or any other issue of a sensitive nature, is addressed in the 1996 Health Insurance Portability and Accountability Act (HIPAA), whose Privacy Rule is enforced by the Office for Civil Rights (OCR). HIPAA protects the privacy of health information while allowing each individual access to their records to see what is written about them and even ask to make corrections to what is documented (Badahman, 2023).
Awareness of their individual rights and the value that each life inherently possesses are crucial foundational steps leading to the inclusion of those suffering from HIV into society and better health. By emphasizing the importance of everyone’s lives, we, as healthcare professionals, can increase participation of the HIV infected individual in their own care and educate them as well.
Important things to educate all individuals about include:
- Keep informed of any changes in HIV knowledge and care
- Use measures to stop the transmission of HIV
- Avoid high-risk behaviors
- Get routine screening for HIV infection, if necessary
- Actively participate in treatment should the individual become infected
Therefore, as we progress to viewing the magnitude of the current HIV pandemic with its slowly widening reach into our culture and homes, remember that we are really dealing with the frightened and wounded. Each is faced with the sudden overwhelming burden of managing the chronic terminal condition of HIV/AIDS.