Presentation of asthma can vary depending on age, severity, and the setting in which symptoms arise, but it typically involves a combination of chronic and acute manifestations.
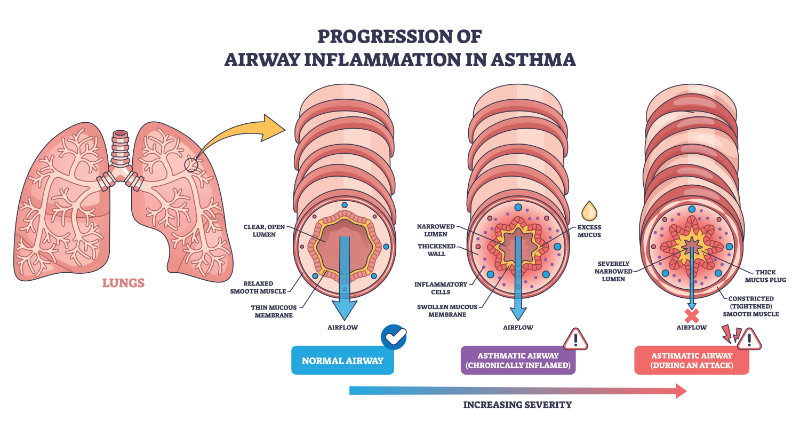
Acute presentation is characterized by sudden exacerbations or “asthma attacks,” during which symptoms become more severe and may include pronounced wheezing, tachypnea, use of accessory muscles for breathing, or difficulty speaking in full sentences. In severe cases, cyanosis, confusion, or “silent chest” are ominous findings that require immediate intervention (Brashers et al., 2025).
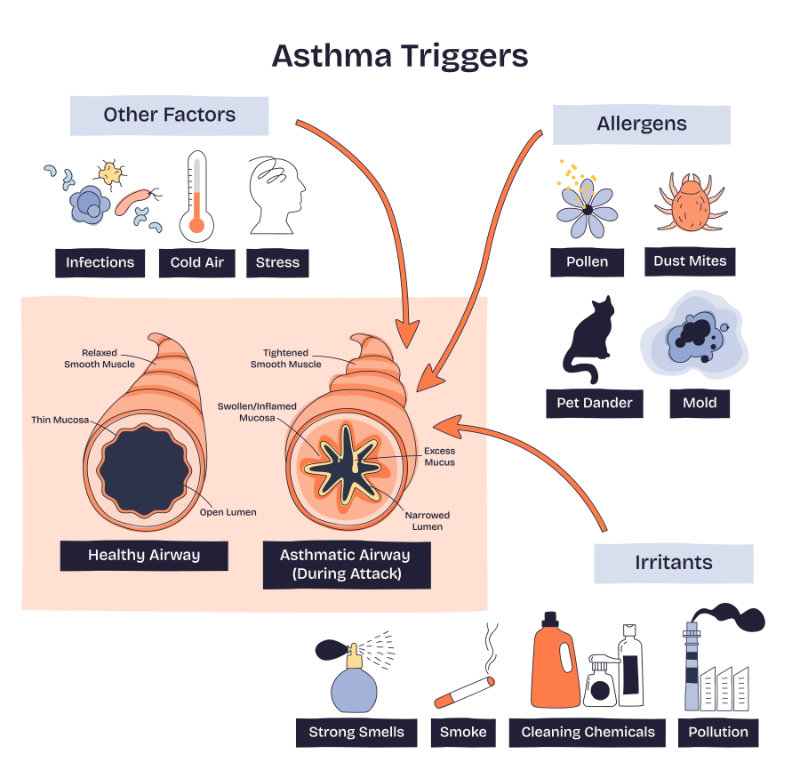
Chronic symptoms often include a persistent or recurrent cough (which may be worse at night or early morning), wheezing, shortness of breath, and chest tightness. These symptoms tend to fluctuate in frequency and intensity, and patients often report that they are triggered by specific exposures such as allergens, respiratory infections, exercise, or changes in weather (Brashers et al., 2025).
In children, asthma may present subtly, especially in its chronic form. Parents often report a recurring cough, particularly at night, after play, or following respiratory infections. Wheezing may be heard intermittently, but in some children, cough is the predominant or only symptom, leading to a diagnosis of “cough variant asthma.” Reports of recurrent episodes of “bronchitis” or “pneumonia” may also suggest undiagnosed asthma. During acute exacerbations, children may present with rapid breathing, retractions, nasal flaring, and difficulty speaking or feeding. Because children may struggle to describe their symptoms, clinicians rely heavily on parental reports of coughing patterns, nighttime awakenings, and response to bronchodilators (Martin et al., 2022).
One useful tool for evaluating asthma in pediatric patients is the Childhood Asthma Control Test (C-ACT), an evidence-based questionnaire designed for children ages 4 to 11. The C-ACT combines the child's self-reported responses (with pictorial response options to aid understanding) and input from the parent or caregiver. Questions focus on the frequency of symptoms such as coughing, wheezing, nighttime awakenings, activity limitations, and the need for rescue inhaler use over the previous four weeks. Each response is scored, and the total score helps determine whether the child’s asthma is well controlled, not well controlled, or poorly controlled. A score of 19 or less generally indicates that asthma is not adequately controlled and may warrant a step-up in therapy, re-evaluation of environmental triggers, or evaluation of treatment adherence. The simplicity and reliability of the C-ACT make it a valuable tool for clinicians, as it provides structured insight into the child’s daily symptom burden and quality of life, supporting both diagnosis and ongoing management (Martin et al., 2022; van Dijk et al., 2020).
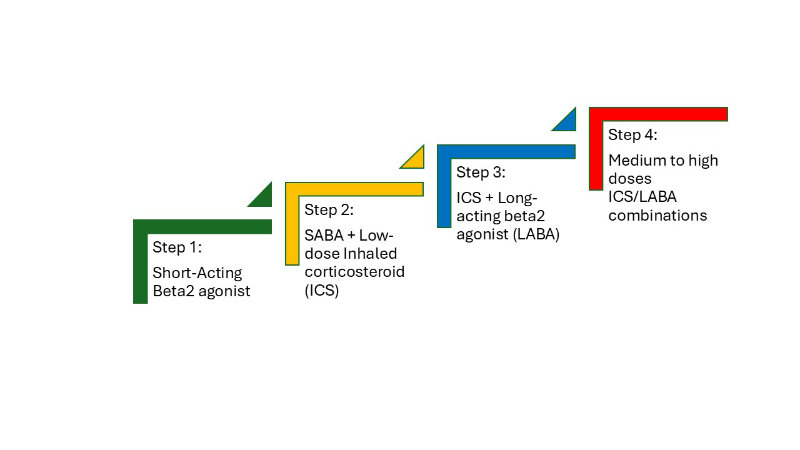
In adults, asthma is often easier to recognize by the pattern of wheezing, chest tightness, and variable breathlessness. Adults may describe episodic symptoms that interfere with sleep, physical activity, or work performance. A thorough history should include questions about occupational exposures (e.g., dust, fumes, or chemicals), smoking status, and environmental triggers at home. Adults with asthma often have comorbidities such as allergic rhinitis, gastroesophageal reflux disease (GERD), or obesity, which can complicate presentation and control. Clinicians assessing adults should document the frequency of daytime and nighttime symptoms, the use of rescue inhalers, and any limitations in daily activities, as this information is key to classifying asthma severity and guiding treatment decisions.
Across all age groups, critical assessment data for clinicians include vital signs (respiratory rate, oxygen saturation, heart rate), auscultation findings (wheezing, decreased air entry), and the presence of accessory muscle use, tripod position, or retractions. History-taking should emphasize symptom variability, triggers, family history of asthma or allergies, and response to previous treatments. Identifying patterns such as worsening symptoms at night, improvement after bronchodilator use, or exacerbations linked to allergens provides strong clues toward the diagnosis. Collectively, these clinical observations form the foundation for confirming asthma with objective testing (Brashers et al., 2025).